Electrochemical Study of a WO₃ NPs/MoO₃ Heterojunction-Based Dual-Enzyme Amperometric Acetylcholine Sensor
Acetylcholine (ACh) is an essential neurotransmitter involved in neural signal transmission, regulating muscle contraction, glandular secretion, brain function, and inflammatory responses. Fluctuations in ACh levels are closely associated with neurodegenerative diseases such as Alzheimer’s and Parkinson’s. The accurate detection of low ACh concentrations is vital for understanding and diagnosing these conditions.
Electrochemical techniques are highly valued for their sensitivity, selectivity, and rapid, cost-effective analysis. The use of flexible printed circuit board (FPCB) electrodes provides precise, scalable, and easily modifiable platforms suitable for a wide range of applications, including catalysis and energy conversion. Recently, modified working electrodes have attracted growing attention in electrochemical ACh detection due to their superior catalytic activity.
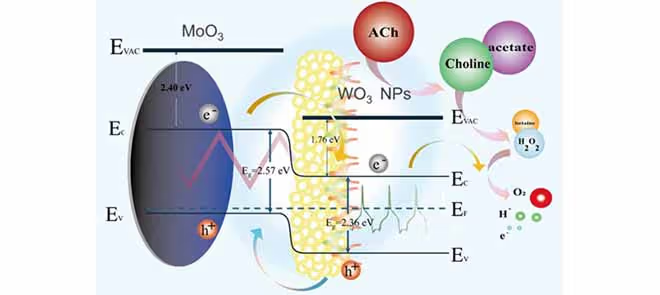
To enhance detection sensitivity, this study proposes a novel current-type amperometric acetylcholine biosensor based on an FPCB, integrating tungsten oxide (WO₃) nanoparticles with molybdenum oxide (MoO₃) thin films to form a heterojunction structure. Current-based sensors were selected for their superior performance in detecting low analyte concentrations. Modified electrodes with oxygen-vacancy-rich MoO₃ films and biocompatible WO₃ nanoparticles exhibited improved ion adsorption, enhanced electron transport, and an enlarged surface area, thereby enhancing electrocatalytic activity.
The MoO₃ thin film was deposited on the FPCB electrode via radio-frequency sputtering, followed by hydrothermal synthesis and drop-casting of WO₃ nanoparticles to form the heterojunction interface.
To improve selectivity, acetylcholinesterase (AChE) and choline oxidase (ChO) were crosslinked into the sensing layer to establish a dual-enzyme catalytic system. The enzymes were dissolved in phosphate-buffered saline (PBS) and immobilized using glutaraldehyde (GA). PBS, which mimics the osmolarity and ionic strength of human body fluids, serves as a nontoxic and isotonic medium for biosensor testing. ACh is hydrolyzed by AChE into choline, which is subsequently oxidized by ChO to produce electrochemically detectable hydrogen peroxide (H₂O₂), generating a current proportional to ACh concentration.
Material characterization using energy-dispersive X-ray spectroscopy (EDX) and atomic force microscopy (AFM) confirmed uniform elemental distribution, high crystallinity of the WO₃/MoO₃ structure, successful enzyme immobilization, and an over 80-fold increase in active surface area. Electrochemical impedance spectroscopy revealed a decrease in charge transfer resistance from 23.87 kΩ to 6.44 kΩ, indicating enhanced electron transport across the heterojunction.
The biosensor exhibited a linear response from 0.01 to 100 µM, with a detection limit of 1.37×10⁻¹⁰ M and a sensitivity of 31.85 µA·decade⁻¹·cm⁻². It demonstrated excellent selectivity against common interferents, including glucose, urea, ascorbic acid, and dopamine, and strong stability at neutral pH. The response time was approximately 6 seconds, and continuous operation remained stable for over 12 days.
These results confirm that the synergistic effect between the WO₃/MoO₃ heterojunction and the dual-enzyme system transforms diffusion-limited detection into an efficient, electron-transfer-dominated response. The proposed biosensor provides a promising, scalable, and low-cost platform for real-time neurochemical monitoring and portable biomedical applications.