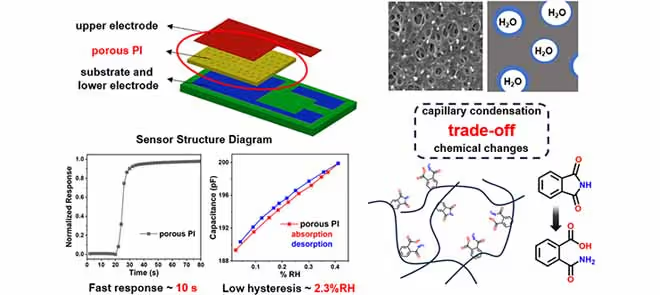
Porous Moisture Sensor Based on NIPS Method: Tradeoff Between Microporous Structure and Chemical Groups to Achieve Low Humidity Hysteresis
Low humidity hysteresis and fast response are core research and development goals for next-generation moisture sensors. However, porous polyimide (PI) sensors fabricated using the nonsolvent-induced phase separation (NIPS) method often exhibit significant hysteresis due to interactions between the sensing material and the nonsolvent coagulation bath. It reduces measurement reliability and limits its applications in various fields, including early fault warning in oil-filled equipment.
To address this critical issue, this study uses ethanol-water solutions with different ethanol mass fractions (0%, 40%, and 80%) as the nonsolvent coagulation bath to fabricate porous PI for moisture sensing. It also compares dense PI to investigate the effect of ethanol mass fraction on the microporous structure parameters and the chemical structure of PI films in NIPS.
The research reveals that the NIPS method reduces the imidization degree of PI while regulating the physical morphology of micropores. Through performance tests and analyses of the impacts of microporous structure parameters and the imidization degree of porous PI films on the moisture sensing performance of the sensor, a design strategy for fast-response, low-hysteresis moisture sensors based on a tradeoff between these two factors is proposed.
Characterization of the physicochemical properties of porous PI films combined with molecular simulation results reveals the core mechanism of this tradeoff strategy: as the ethanol mass fraction increases from 0% to 80%, the pore size of PI films gradually decreases from the micrometer scale to the hundreds of nanometer scale.
While the formation of a microporous structure can increase the specific surface area, accelerate water molecule adsorption, and improve response speed, excessively small pores can trigger the capillary condensation effect, trapping water molecules on pore walls, and making them difficult to desorb, thereby increasing hysteresis.
However, an overly low ethanol mass fraction reduces the imidization degree of PI (the imidization degree of films prepared with pure water is only about 50%), which introduces hydrophilic groups and decreases the free volume fraction. This not only enhances the interaction between PI and water molecules but also restricts the diffusion of water molecules, ultimately leading to increased hysteresis. Therefore, an appropriate ethanol mass fraction must be selected to balance the effects of micropore size and the degree of PI imidization.
Performance tests demonstrate that based on the tradeoff strategy between microporous structure and chemical groups, the sensor fabricated with a 40% ethanol solution achieves an optimal performance balance, combining a fast response (9s) and low humidity hysteresis (2.8%RH) with a sensitivity of approximately 0.3 pF/% RH and excellent stability in oil. Its comprehensive performance ranks among the forefront of existing research, providing an important reference for the design of low-hysteresis and fast-response humidity sensors.
The study also reveals that, when using the NIPS method to prepare porous materials (not limited to PI), in addition to the microporous structure, future research should also consider changes in the chemical composition of the sensing material and their impacts on material performance and applications.