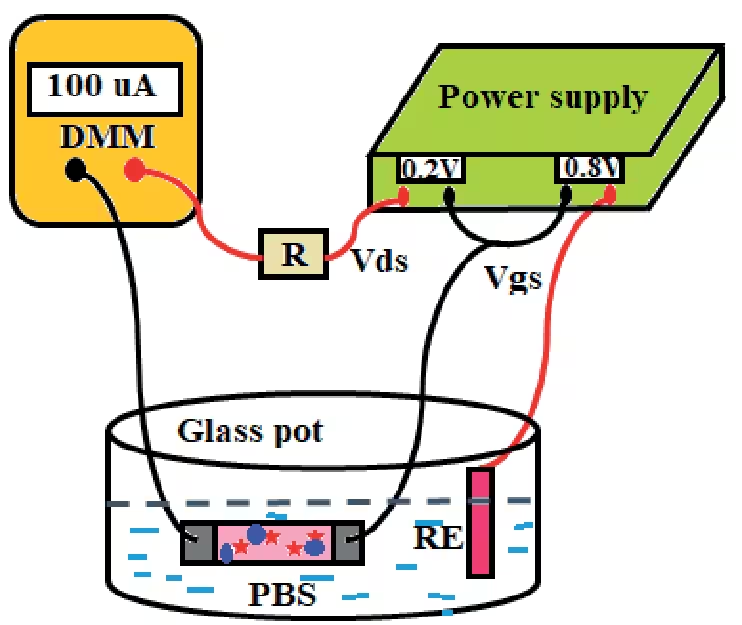
Highly Sensitive Enzyme-Modified Field Effect Transistor Based Biosensor for Sarcosine Detection
Ion-Sensitive Field Effect Transistor (ISFET) is an electronic device that measures the gate insulator-electrolyte interface potential i.e., surface potential. Enzyme-modified FET (ENFETs) is formed by coupling the surface of an ISFET with enzymes. In ENFET, the change in surface potential of ISFET is caused by a catalytic reaction (between the enzyme and its substrate). The change in surface potential leads to a modulation of the channel conductance and, consequently, the drain current of the ENFET. Thus, by measuring changes in the drain current, the concentration of the analyte under examination can be determined quantitatively.
Sarcosine, a non-proteinogenic amino acid, has been considered as an effective Prostate Cancer biomarker. Early detection of cancer can increase the survival rate from 10% to 90%. Despite the state-of-the-art devices and conventional methods developed for its detection, key challenges remain, mainly due to the high limit of detection (LOD), poor electron transfer characteristics, the lack of real-time monitoring, and the presence of interferents.
To address the challenges, in this study, an ENFET based biosensor has been fabricated by solution mixing method where indium tin oxide (ITO) coated glass slide has been used as FET substrate, polyethyleneimine-doped carboxylic acid functionalized multiwalled carbon nanotube (PEI-doped COOH-WMCNT) is used as transporting channel and, silver metal has been used for fabrication of source and drain regions by physical vapor deposition (PVD) technique. Niobium pentoxide (Nb2O5), having a high dielectric constant value of ~41 is used as the Insulating layer.
Dimethyl sulphoxide-treated poly(3,4-ethylenedioxythiophene): polystyrene sulfonate-multi-walled carbon nanotube-chitosan (DMSO treated PEDOT: PSS-MWCNT-CHIT) has been used as a supporting layer for immobilization of the enzyme sarcosine oxidase (SOx).
The successful synthesis, deposition, uniformity, and conductivity of the layers were confirmed through characterization methods such as Fourier transform infrared spectroscopy (FTIR), Powder X-ray diffraction (PXRD), Scanning electron microscopy (SEM), transmission electron microscopy (TEM), Atomic force microscopy (AFM), Cyclic voltammetry (CV), Amperometry, and Electrochemical Impedance Spectroscopy (EIS).
The integration of nanomaterials in sensor development has revolutionized biosensing technology, enabling accurate disease diagnosis for use in the healthcare sector. One-dimensional nanomaterials (1Ds), mainly carbon nanotubes (CNTs), possess high electrical conductivity, stability, large surface area, and, on integrating with high-k dielectric materials, alter their covalent bonds due to p-p interactions.
The reinforcement of CNT with polymers increases the sensing performance of the biosensor, as it selectively binds to the target analyte of interest. Tin-doped indium oxide (ITO) coated glass is used as the alternative electrode to gold, silver, or platinum electrodes, mainly due to its high conductivity, low cost, and biocompatibility.
The experimental results demonstrated that the biosensor shows superior performance characteristics for detecting sarcosine with very high current and voltage sensitivity 3.43x103 μA/μM and 64 mV/log[molar], very low limit of detection 0.963 nM, acceptable stability of four months in the linear range 0.89-123.846 μM, excellent correlation (R2 = 0.9905), along with good repeatability and reproducibility.
The fabricated CNT-ENFET-based biosensor, therefore, acts as a cost-effective, non-invasive, promising candidate for quick, sensitive, and accurate detection of very low concentrations of sarcosine, aiding in early disease diagnosis and monitoring.