A Terahertz Split Ring Resonator Nanosensor for Cardiac Biomarker Detection
Cardiac disease remains one of the leading causes of death worldwide and has even become quite common among youngsters. An early diagnosis of a heart condition can save lives. Cardiac biomarkers, such as proteins released during a heart attack, are critical for diagnosing cardiac dysfunction, particularly heart failure. Laboratory tests, while reliable, require specialized lab equipment, trained professionals, and time. In an emergency, time is crucial for saving lives.
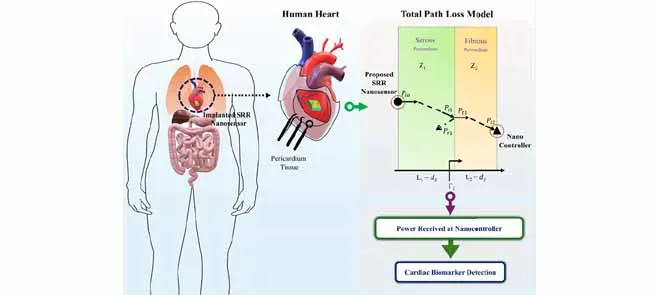
This paper introduces a miniature biosensing platform that integrates an ultrasensitive terahertz (THz) metamaterial sensor with an intrabody THz communication link model, specifically designed for the heart’s outer covering, the pericardium. Though cardiovascular disease remains a leading cause of mortality, most existing monitoring approaches operate at a macroscopic level, relying on wearable devices that infer cardiac condition indirectly rather than directly detecting biochemical changes near the heart. The proposed system addresses this gap by enabling localized molecular sensing around cardiac tissue and facilitating wireless signal transmission to a nearby controller.
In this work, the sensing element is a terahertz split-ring resonator (SRR), a subwavelength metallic structure engineered to exhibit a sharp resonance at a specific THz frequency. SRRs can concentrate electric fields within their capacitive gaps, creating localized “hot spots” that are highly sensitive to changes in the surrounding dielectric environment.
The target analyte is a clinically recognised cardiac stress biomarker, N-terminal pro–B-type natriuretic peptide (NT-proBNP). The concentration of NT-proBNP increases under pressure or volume overload conditions in the heart. When NT-proBNP binds to a functionalized SRR surface, it alters the local permittivity, resulting in a measurable shift in the resonance frequency.
A double-ring SRR nanosensor is designed on a biocompatible polydimethylsiloxane (PDMS) substrate using gold metallization and optimized through full-wave electromagnetic simulations. The design achieves a strong absorption peak near 0.867 THz with a narrow linewidth, supporting high detection precision. An equivalent circuit model developed in Advanced Design System (ADS) provides physical insight into how biomolecular binding modifies the effective capacitance and shifts the resonance.
Sensing performance is evaluated by varying the thickness and refractive index of analytes that resemble protein-like materials. In both cases, increasing thickness or refractive index results in a systematic shift in the resonance frequency, consistent with an increase in effective permittivity. The sensor demonstrates a peak sensitivity of 1460 GHz/RIU(Refractive Index Unit), a quality factor of approximately 22, and a strong figure of merit, comparing favorably with previously reported THz biosensors.
The paper introduces an intrabody channel model for THz signal propagation through the serous and fibrous layers of the pericardium. Using transmission-line theory, the model accounts for factors such as tissue impedance, reflections at layer interfaces, absorption, and spreading loss. This allows for estimating the power received by an implanted nanocontroller with analytical results validated using COMSOL simulations.
By integrating biomolecular sensing with intrabody signal propagation, this research transcends isolated sensor optimization. It establishes a system-level framework for THz-based wireless, implantable, faster, and more accessible cardiac diagnostic testing. This innovation could lead to the early diagnosis and treatment of heart disease.