Precise Detection and Quantitative Prediction of Blood Glucose Level With an Electronic Nose System
Diabetes is a common condition that affects millions worldwide in all age groups. Monitoring glucose levels is crucial but arduous due to blood sampling and needs urgent improvement. The current "Non-invasive" wearables are costly and partially reliant on blood interaction. Scientists have successfully developed an electronic nose (E-nose), a sensing system consisting of a variety of gas sensors to diagnose different respiratory diseases.
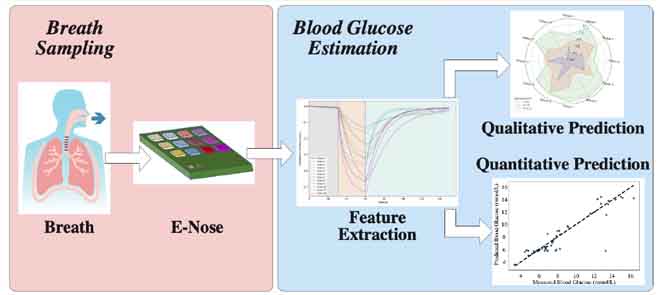
The researchers have designed and evaluated an E-nose system in conjunction with machine learning (ML) models to identify blood glucose levels by analyzing the exhaled breath. The concentration of acetone in exhaled breath is a key biomarker for diabetes. The prototype uses ML classifiers and regressors to predict blood glucose levels qualitatively and quantitatively using the data from the E-nose sensor.
The E-nose system employed an array of 12 gas sensors within a gas chamber for breath analysis. A customized signal conditioning circuit captured and transmitted sensor signals to smartphones via Bluetooth. The data were saved to a cloud server for further processing. A web interface was used for the display of data and results.
The researchers prepared the dataset for training consisting of breath samples from 41 participants for five continuous days. The participants were instructed to exhale into a Tedlar sample bag after regular fasting. Before the collection of breath samples, the glucose levels of all participants were also measured via a blood test for calibration and parameter training. Participants' glucose levels were classified into three ranges (below 6.0 mmol/L, 6.0-7.9 mmol/L, and above 7.9 mmol/L).
To minimize temperature variations, the breath sample bags are brought to room temperature while the E-nose chamber is maintained at 60°C. Each 150s measurement cycle comprised a baseline, response, and recovery phase. Before the breath sample inhalation, the chamber and sensors are stabilized to baseline. After reading the sensor outputs during the response phase, the system is exhausted by injecting clean air during the recovery phase.
The sensor signals in response to gases in samples are transformed into feature vectors. To avoid model overfitting caused by complex feature extraction, maximum responses from each measurement were used as features for glucose prediction modeling.
The researchers employed three frequently based classifiers: support vector machine (SVM), random forest, and gradient boosting tree (GBT) for qualitative analysis. Similarly, SVM regressor (SVR), random forest regressor, and GB regressor (GBR) were used for quantitative glucose prediction. GBR offered the best prediction performance (0.77 mmol/L MAE). The researchers have created a hybrid regressor that merges GBT classification with either SVR or GBR regression techniques, significantly enhancing prediction accuracy. The hybrid regressor first qualitatively predicted the range of glucose levels and generated fine-grained quantitative predictions based on the predicted range. The method significantly improves prediction accuracy (0.69 mmol/L MAE) by addressing the imbalanced dataset issue.
The machine learning-enabled E-Nose system demonstrated accurate glucose measurement capability non-invasively. The prototype offers promising possibilities for enhancing healthcare, particularly for individuals with diabetes who require frequent glucose monitoring.