Miniaturized System for Tumor Cell Detection and Differentiation
Research has been going on for decades for detecting and differentiating circulating Tumor Cells (CTC) as part of liquid biopsy for early, non-invasive diagnosis and prognosis of the disease. CTCs are cells released into the circulation by malignant tumors. CTC detection is often done by capturing and enriching (isolating) cells from blood samples. Using fluorescence microscopy, molecular, and impedance analyses, the separated cells are then distinguished from non-tumor cells such as leukocytes. EpCAM and PSMA are currently utilized as standard CTC membrane antigens.
Electrical impedance spectroscopy (EIS) is another emerging cell detection technology. The behavior of cells may be characterized and discriminated using data on inductance, capacitance, and impedance. The EIS sensor works on the principle of cell polarization caused by an electric field, which, in turn, causes a change in electrical impedance, resulting in quantitative CTC detection.
The medical fraternity has not widely accepted most available EIS and non-EIS devices as they require expensive, bulky and specialized instruments and trained operators.
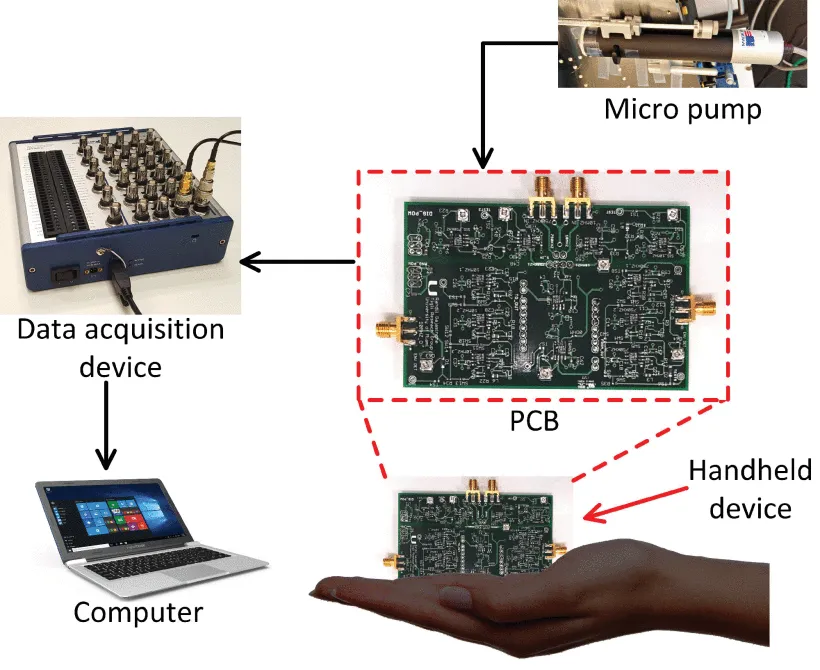
The researchers created and tested an automated, non-invasive, and compact system for detecting and differentiating cultivated tumor cells. A micro-fabricated EIS sensor, a data acquisition device (DAQ), a customized printed circuit board (PCB) and software with a simplified graphic interface and automated analysis make up the system.
The EIS sensor fabrication process is simple and utilizes standard micro-fabrication materials and processes. The sensor chip, fabricated on silicon wafers, consists of a microfluidic channel etched in silicon with gold semi-parallel channel sidewalls as micro-electrodes. A photolithography process is initially utilized to create the microfluidic channel features on the wafer, followed by an etch mask containing an oxide layer. Anisotropic etching of silicon in heated KOH is then used to create the channel. Wet thermal oxidation is used to eliminate the leftover oxide layer and build a new insulating layer. The micro-electrode sensors are formed using gold and AZ nLOF 2020 photoresist. Finally, the microfluidic channel is sealed using a reversible Polydimethylsiloxane (PDMS) layer.
A Printed Circuit Board (PCB) was customized for generating excitation signals, signal conditioning and impedance measurements at two frequencies.
When an excitation voltage is applied between the electrodes, small current flows through the cell medium filled in a channel. This current is inversely proportional to the impedance between electrodes. A cell passing between electrodes increases channel impedance. This change in channel impedance is ultimately reflected as negative peaks in output voltage. A DAQ converts voltage readings to digital information by sampling output voltage at high and low frequencies. The digital data is then processed to remove background noise, normalized and plotted on a scatter plot.
Two tumor cells and a normal cell from the same source were used to test this cell detection technique. These cells were grown to assure a sample of primarily single cells before being discharged into the EIS chip's input reservoir. The connected software automatically processed the sampled data. The healthy cell line was clearly distinguished from the two tumor cell lines on the scatter plot.
The technology demonstrated here can be enhanced to become a low-cost, portable, and automated CTC detection device. The PCB can be configured to probe various frequency bands and electrode pairs simultaneously. Data processing methods that improve differentiation accuracy must be developed.