Investigation on Butanone Sensing Properties of ZnO Sensor Under Different Calcination Temperature
Butanone, a colorless and almost odorless chemical, is widely used in industrial productions and interior decoration of vehicles. It is highly flammable, toxic, carcinogenic, and adversely affects human health on long exposure or inhalation. It is essential to employ highly sensitive and selective butanone sensors for early detection and warning.
Metal oxide semiconductor (MOS)-based butanone sensors have high response characteristics and low detection limit. These low-cost and small sensors detect butanone through a simple process. Sensors based on noble metal-modified nanomaterials composites exhibit high biocompatibility and catalytic activities and have overall structural robustness. However, they show low sensitivity and selectivity in detecting different substances.
A zinc oxide (ZnO) gas sensor shows high sensitivity with a low ppb detection level and can detect gases in different environments. ZnO nanomaterials can be conveniently synthesized and morphed as rods, spheres, and flowers by hydrothermal method.
The researchers present a comprehensive study based on experimental analysis of the effect of calcination temperature on the overall performance of a ZnO gas sensor. The particle size of mesoporous-ZnO (M-ZnO) was regulated by adjusting the calcination temperature. The gas sensing performance of the material was investigated for varying particle sizes.
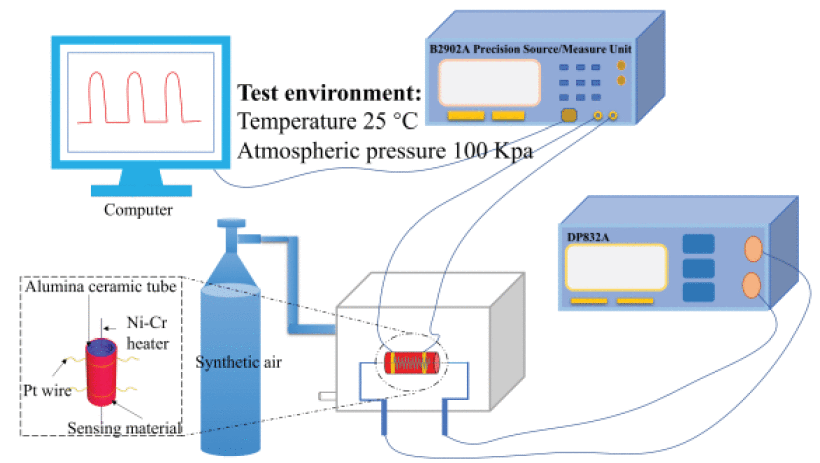
Samples of M-ZnO-350, M-ZnO-400, and M-ZnO-500 were obtained by annealing the ZnO powder at 350°C, 400°C, and 500°C. Butanone sensing properties of all samples were tested for response in the concentration range of 0.2 ppm to 100 ppm butanone. The particle size of the samples was controlled by the calcination procedure.
The particle size of the materials first decreased and then increased with the increase in calcination temperature. Material response values were found to be consistent with particle size. The particle size was the smallest when calcinated at 400°C, and the gas sensitivity improved significantly.
The M-ZnO sensor response to 100 ppm of butanone at the optimum operating temperature was found to be highest when the particle size was close to 2 times the Debye length. The M-ZnO-400 sensor’s response to 151 ppm of butanone had a detection limit of 200 ppb, with a 4.5 s response time and 5 s recovery time. This response is at least 62 times higher than other gases found in car exhaust.
The density functional theory (DFT) calculation was used to simulate the target gases for bond population analysis. Calculations showed that the band gap width of ZnO became smaller with butanone adsorption. The low population value of the C-C bond in the butanone makes it unstable. The oxidization and transfer of electrons on the surface of the nanomaterial lead to the shrinking of the electron depletion layer and a decrease in resistance.
The study shows that the sensing response of the nanomaterial to butanone is directly related to its particle size. The smaller the particle size of the material, the higher the response. The sensing feature of nanomaterials can be improved by regulating the particle size through calcination. With experimental analysis of sensor sensitivity and selectivity, the study provides an effective design template for high-performance butanone sensors.